
Sampling Assemblies

Sampling Assemblies
Sampling is a critical step in bioprocess operations ,enabling process monitoring, quality control, and regulatory compliance without compromising sterility. Our sampling assemblies ensure representative, aseptic sample collection with minimal impact on process integrity.
5 Port Needle Holder
The 5-port needle holder allows simultaneous, sterile access to multiple lines, simplifying sampling and fluid transfer from SS tanks. Its ergonomic design ensures easy handling, while precision-engineered ports guarantee leak-proof, closed-loop operation, minimizing contamination risk in complex bioprocess workflows.

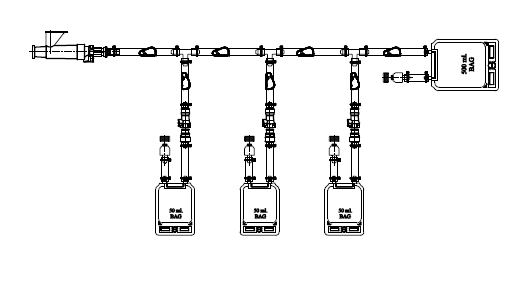
Needle Sampling Manifold Assembly

Inteviz AQG sampling manifold assembly
AQG sampling manifold assembly designed for sterile, closed-loop sampling from single-use bags and tanks—simple, secure, and reliable.
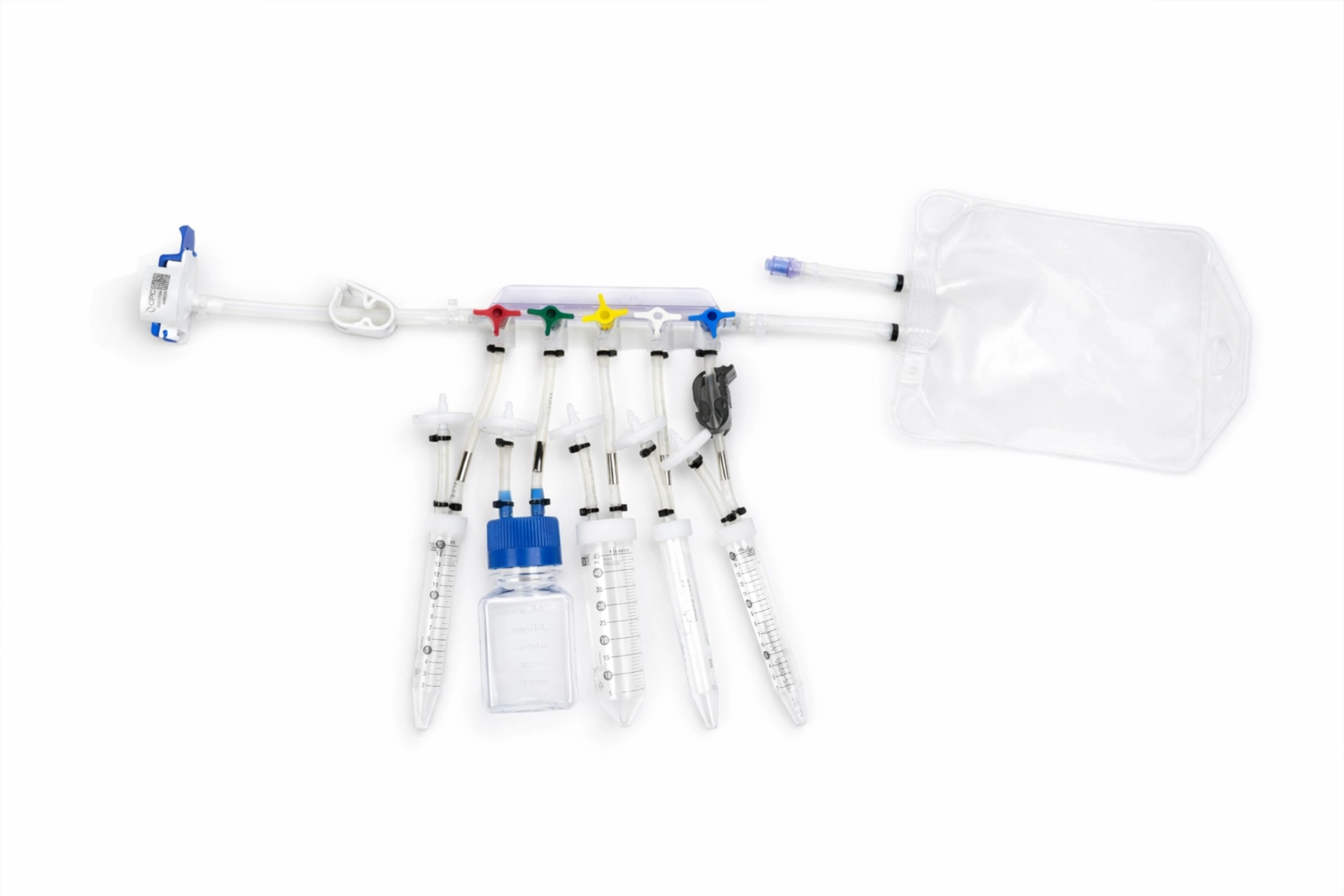
Inteviz STC Sampling Assemblies
Our STC Sampling Assembly provides a simple and user-friendly solution for sterile sampling in bioprocess applications. Designed for easy integration with SS tanks, it supports closed-loop operation, reduces handling errors, and ensures leak-proof, contamination-free sampling. STC sampling Assembly
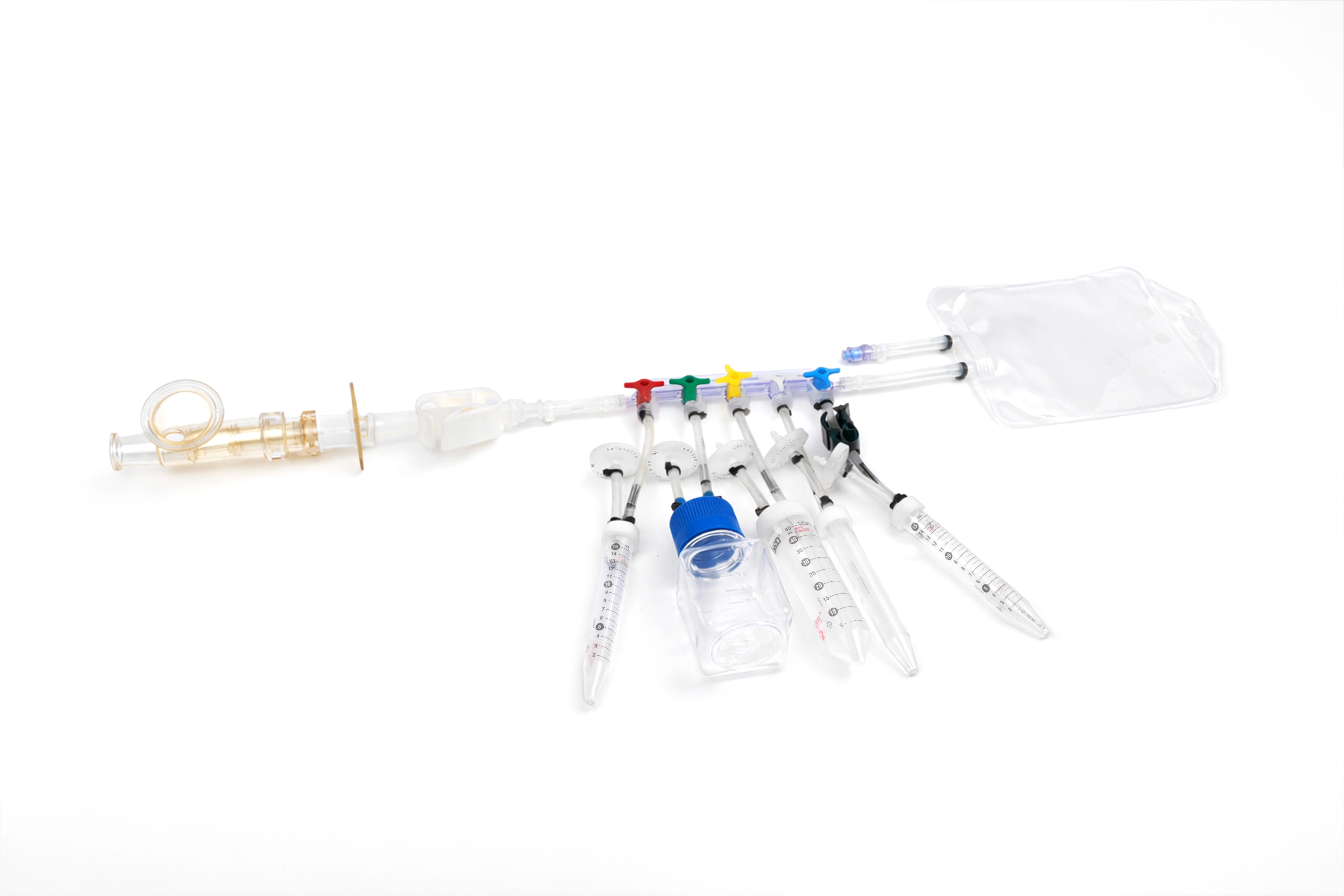
Bottle needle sampling Assembly

Tube needle sampling assembly

Bag needle sampling assembly
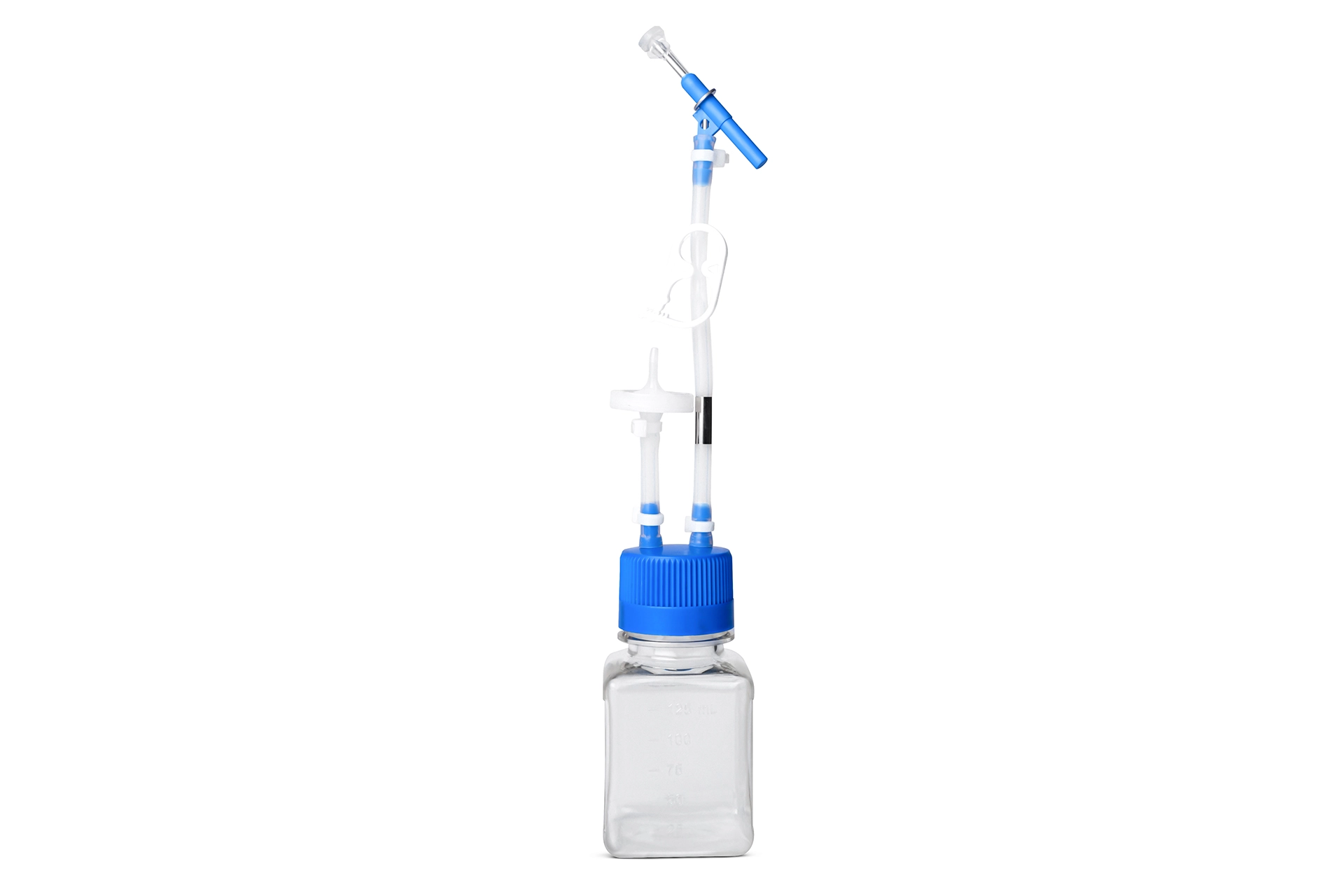
Sampling Manifold assembly
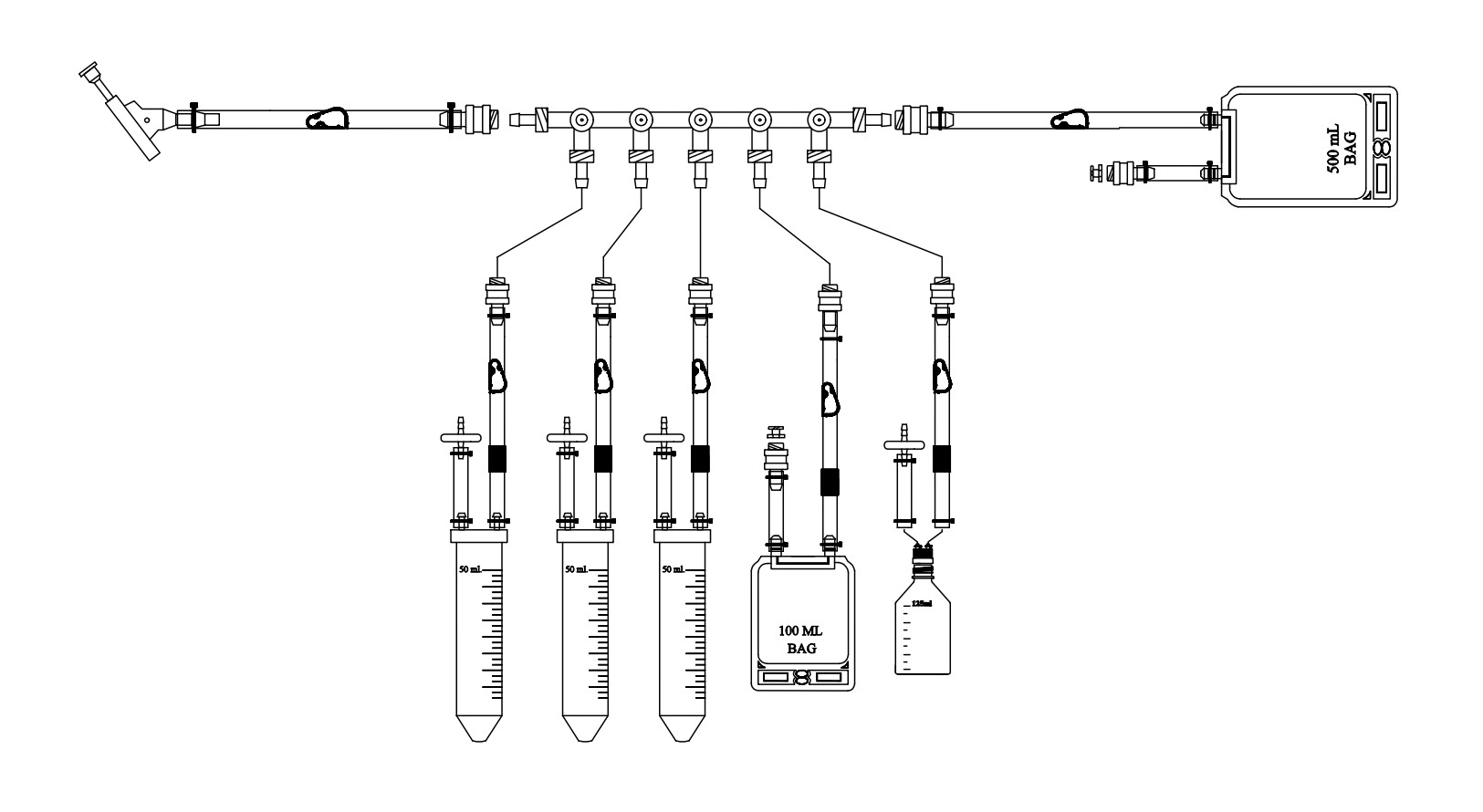
Sampling Manifold assembly
Customization Capabilities
Sampling assemblies can be customized for STC, S2C, and AQG sampling with flexible port configurations, tubing sizes, materials, and connection interfaces. Designed for single-use bags, tanks, and SS systems, each assembly supports closed-loop operation, ease of handling, and reliable, contamination-free sampling.
Customization Capabilities
Sampling assemblies can be customized for STC, S2C, and AQG sampling with flexible port configurations, tubing sizes, materials, and connection interfaces. Designed for single-use bags, tanks, and SS systems, each assembly supports closed-loop operation, ease of handling, and reliable, contamination-free sampling.
Every Inteviz Assembly is backed by our expertise in sterile single-use manufacturing and deep understanding of bioprocess needs. From media preparation to final filling, we ensure your fluid handling is secure, efficient, and regulatory-ready.
|
Parameter
|
Value / Option
|
|---|---|
|
Bottle Volume
|
60 mL to 2000 mL
|
|
Material
|
PC, PETG, HDPE, PP
|
|
Tubing Options
|
C-Flex®, TPE, Silicone (various IDs)
|
|
Connector Compatibility
|
Luer, MPC, AseptiQuik, Dip Tubes
|
|
Sterilization
|
Gamma irradiated
|
|
Certification
|
USP , , ISO 10993
|
Applications
Upstream Bioprocessing
-
Media & buffer preparation
-
Seed train expansion
-
Bioreactor feeding & harvesting
-
Closed in-process sampling transfer
Markets Served: Vaccine, Biologics, Cell & Gene Therapy, MAB ,Oncology,API, CDMO
Downstream Purification
-
Chromatography feed & collection
-
TFF, depth filtration, sterilizing filtration
-
Buffer exchange & intermediate hold
Markets Served: Biologics ,MAB ,Oncology ,Vaccine ,Cell & Gene Therapy ,API ,CDMO
Formulation & Final Bulk Handling
-
Bulk drug substance (BDS) transfer
-
Formulation blending
-
Bulk dispensing to final containers
Markets Served: Vaccine ,Biologics ,MAB ,Oncology ,Formulation / Fill Finish ,API ,CDMO
Sterile Fill–Finish Operations
-
Vial, syringe & cartridge filling
-
Multi-line dispensing assemblies
-
Isolator & RABS integrations
-
Precision final dosing
Markets Served: Vaccine ,Biologics ,MAB ,Oncology ,Cell & Gene Therapy ,Formulation / Fill Finish ,CDMO
Sampling & Quality Control
-
Sterile in-process sampling
-
QC / Microbiology sampling
-
Chain-of-custody closed transfer
Markets Served: Biologics ,Cell & Gene Therapy ,Vaccine MAB ,Oncology ,API ,CDMO
Cell & Gene Therapy Workflows
-
Viral vector handling (AAV, Lenti)
-
Closed small-volume transfers
-
Cryo-bag filling & aliquoting
Markets Served: Cell & Gene Therapy ,Biologics ,Oncology ,CDMO
Vaccine Manufacturing
-
Antigen preparation
-
Buffer exchange
-
Final vaccine filling lines
Markets Served: Vaccine ,Biologics ,Formulation / Fill Finish ,CDMO
Quality Built into Every Step
From sourcing globally trusted components to final cleanroom assembly and post-production testing, every step is governed by rigorous quality protocols. Our cleanroom environment, documentation control, and traceability systems ensure our customers receive products that are safe, consistent, and compliant every single time.

