
Sensor Assembly

Sensor Assembly
Our Inteviz Sensor Assemblies are customised, ready-to-use single-use solutions engineered for real-time process monitoring with exceptional accuracy. Powered by world-class pressure, temperature, conductivity, pH, and flow sensors, these assemblies integrate seamlessly into your bioprocess setup while ensuring complete sterility and true plug-and-play convenience. With Inteviz, you gain precise, reliable data—enabling smarter control, higher consistency, and confident decision-making across every stage of your bioprocess.
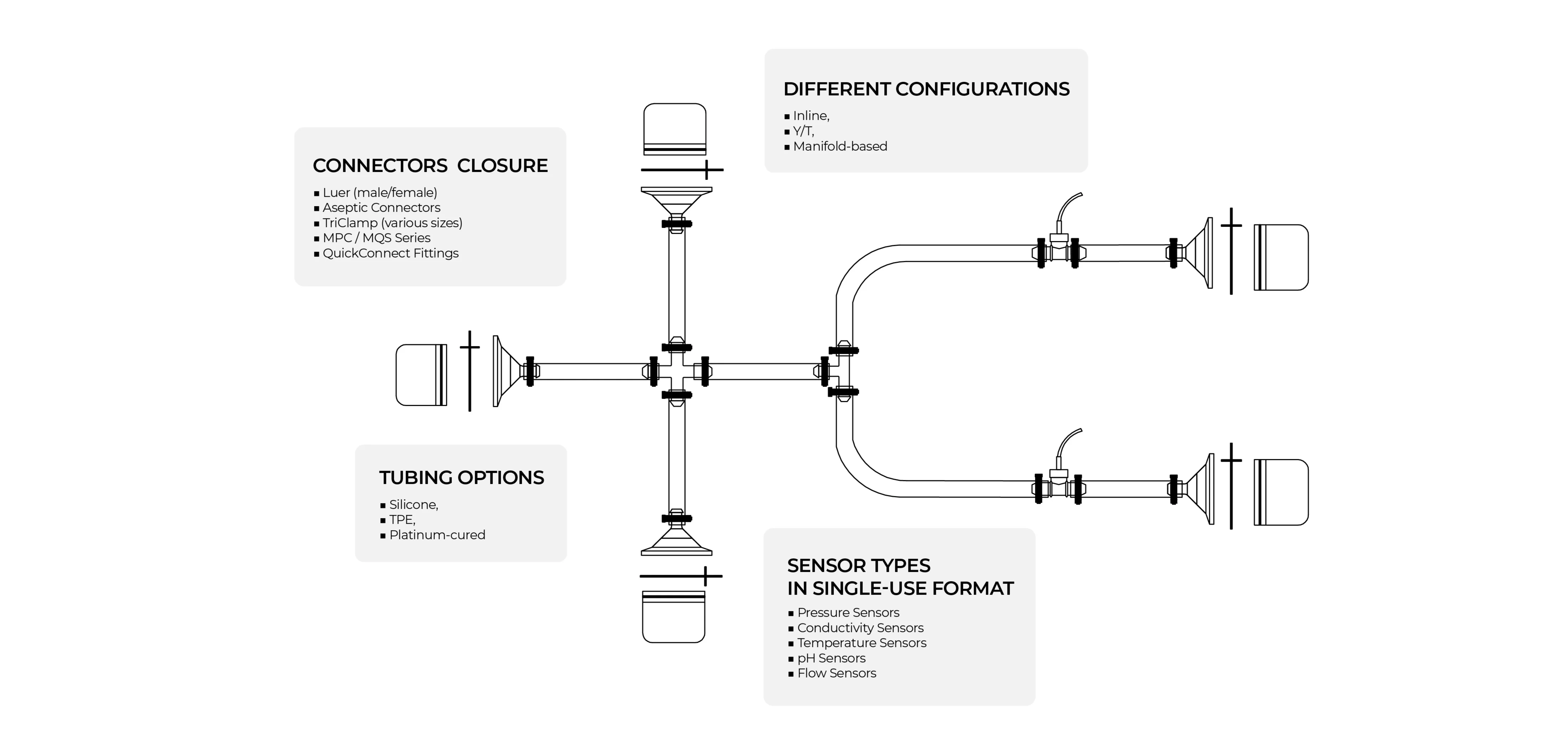
Customization Capabilities
Our Inteviz single-use Sensor Assemblies are fully customizable for precise bioprocess monitoring. You can configure the number of sensors and the required sensor types—whether for pressure, conductivity, temperature, pH, or flow—along with the appropriate tubing, connectors, and custom port layouts. Optional add-ons such as clamps, holders, and sampling ports can also be included to enhance process control. Each assembly is available in sterile, ready-to-use or double-bag packaging, ensuring seamless integration into various listed bioprocessing workflows.
Customization Capabilities
Our Inteviz single-use Sensor Assemblies are fully customizable for precise bioprocess monitoring. You can configure the number of sensors and the required sensor types—whether for pressure, conductivity, temperature, pH, or flow—along with the appropriate tubing, connectors, and custom port layouts. Optional add-ons such as clamps, holders, and sampling ports can also be included to enhance process control. Each assembly is available in sterile, ready-to-use or double-bag packaging, ensuring seamless integration into various listed bioprocessing workflows.
Applications
Upstream Bioprocessing
-
Media & buffer preparation
-
Seed train expansion
-
Bioreactor feeding & harvesting
-
Closed in-process sampling transfer
Markets Served: Vaccine, Biologics, Cell & Gene Therapy, MAB ,Oncology,API, CDMO
Downstream Purification
-
Chromatography feed & collection
-
TFF, depth filtration, sterilizing filtration
-
Buffer exchange & intermediate hold
Markets Served: Biologics ,MAB ,Oncology ,Vaccine ,Cell & Gene Therapy ,API ,CDMO
Formulation & Final Bulk Handling
-
Bulk drug substance (BDS) transfer
-
Formulation blending
-
Bulk dispensing to final containers
Markets Served: Vaccine ,Biologics ,MAB ,Oncology ,Formulation / Fill Finish ,API ,CDMO
Sterile Fill–Finish Operations
-
Vial, syringe & cartridge filling
-
Multi-line dispensing assemblies
-
Isolator & RABS integrations
-
Precision final dosing
Markets Served: Vaccine ,Biologics ,MAB ,Oncology ,Cell & Gene Therapy ,Formulation / Fill Finish ,CDMO
Sampling & Quality Control
-
Sterile in-process sampling
-
QC / Microbiology sampling
-
Chain-of-custody closed transfer
Markets Served: Biologics ,Cell & Gene Therapy ,Vaccine MAB ,Oncology ,API ,CDMO
Cell & Gene Therapy Workflows
-
Viral vector handling (AAV, Lenti)
-
Closed small-volume transfers
-
Cryo-bag filling & aliquoting
Markets Served: Cell & Gene Therapy ,Biologics ,Oncology ,CDMO
Vaccine Manufacturing
-
Antigen preparation
-
Buffer exchange
-
Final vaccine filling lines
Markets Served: Vaccine ,Biologics ,Formulation / Fill Finish ,CDMO
With years of bioprocessing expertise, Shah Brothers delivers locally assembled solutions built on globally trusted technologies — ensuring faster lead times, robust quality and customised configurations for every application.
Comprehensive documentation support available, including BSE/TSE statements, gamma sterilisation certificates and validation dossiers.
|
Parameter
|
Value / Option
|
|---|---|
|
Assembly Type
|
Single-Use
|
|
Sensor Compatibility
|
Hamilton, PreSens, PendoTECH, etc.
|
|
Tubing Size Options
|
1/8” to 1” (custom on request)
|
|
Connector Types
|
Luer lock, CPC AseptiQuik, weldable
|
|
Sterilization
|
Gamma irradiated
|
|
Assembly Validation
|
100% leak tested and traceable
|
Quality Built into Every Step
From sourcing globally trusted components to final cleanroom assembly and post-production testing, every step is governed by rigorous quality protocols. Our cleanroom environment, documentation control, and traceability systems ensure our customers receive products that are safe, consistent, and compliant every single time.

