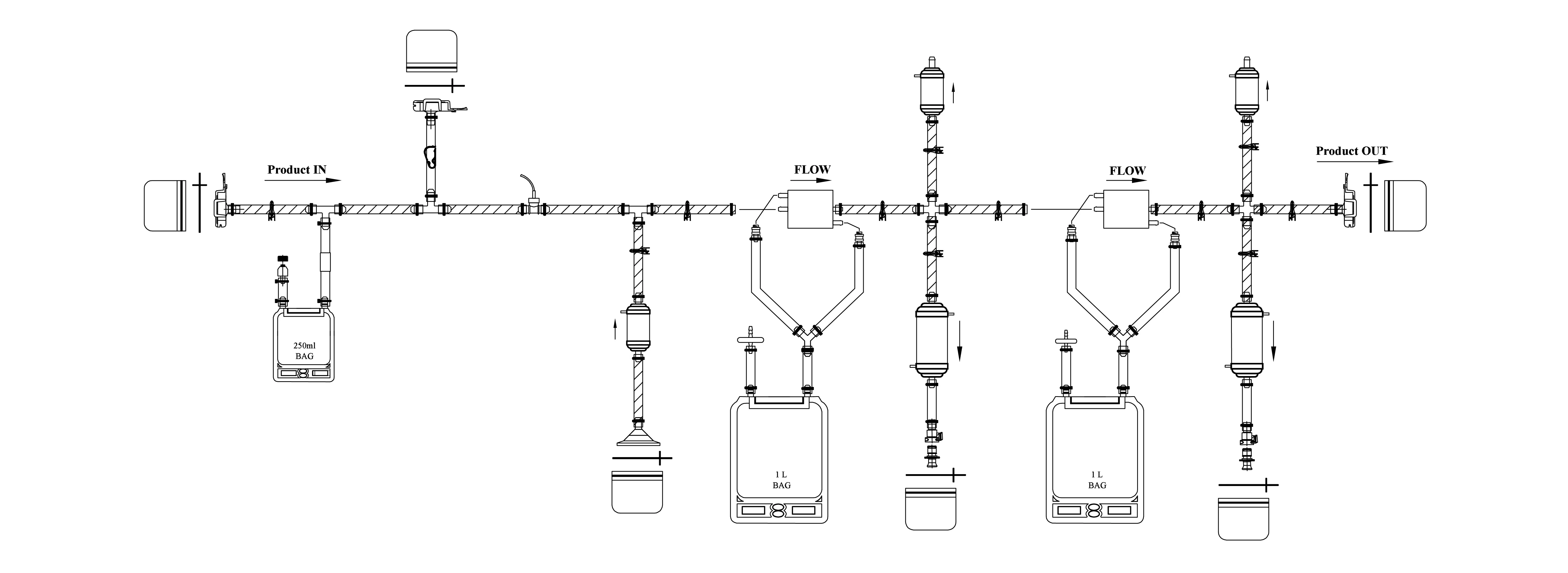
Filter Assembly

Filter Assembly
Our Inteviz Filter Assemblies are engineered with a deep understanding of how critical filtration is to your bioprocess. Built for sterility, scalability, and consistent performance, our single-use filter assemblies ensure secure, convenient, and flexible operations you can rely on. Whether you’re reducing bioburden or achieving final sterilization, Inteviz delivers precision-driven solutions, so you can stay focused on what matters most: advancing life-saving science.
We also custom-design PUPSIT assemblies to your specific needs, with integrated test ports, vent lines, and optimized tubing for safe pre-use post-sterilization integrity testing. Each assembly is delivered sterile and ready-to-use, ensuring seamless integration into Inteviz biopharma filtration workflows.
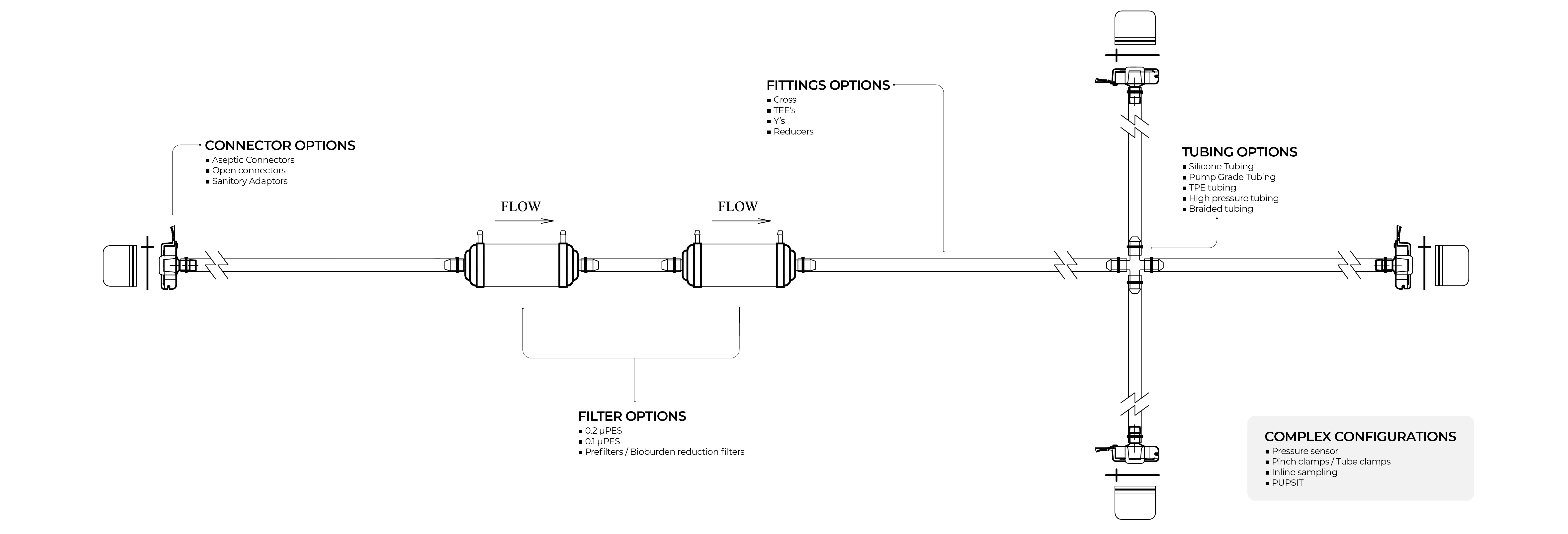
Customization Capabilities
Our Inteviz Filter Assemblies offer maximum flexibility across upstream, downstream, and final filtration applications. You can submit your own filters or select from the Shah Brothers library, and we guide you in choosing tubing, connectors, and port orientations.
Customization Capabilities
Our Inteviz Filter Assemblies offer maximum flexibility across upstream, downstream, and final filtration applications. You can submit your own filters or select from the Shah Brothers library, and we guide you in choosing tubing, connectors, and port orientations.
Applications
Upstream Bioprocessing
-
Media & buffer preparation
-
Seed train expansion
-
Bioreactor feeding & harvesting
-
Closed in-process sampling transfer
Markets Served: Vaccine, Biologics, Cell & Gene Therapy, MAB ,Oncology,API, CDMO
Downstream Purification
-
Chromatography feed & collection
-
TFF, depth filtration, sterilizing filtration
-
Buffer exchange & intermediate hold
Markets Served: Biologics ,MAB ,Oncology ,Vaccine ,Cell & Gene Therapy ,API ,CDMO
Formulation & Final Bulk Handling
-
Bulk drug substance (BDS) transfer
-
Formulation blending
-
Bulk dispensing to final containers
Markets Served: Vaccine ,Biologics ,MAB ,Oncology ,Formulation / Fill Finish ,API ,CDMO
Sterile Fill–Finish Operations
-
Vial, syringe & cartridge filling
-
Multi-line dispensing assemblies
-
Isolator & RABS integrations
-
Precision final dosing
Markets Served: Vaccine ,Biologics ,MAB ,Oncology ,Cell & Gene Therapy ,Formulation / Fill Finish ,CDMO
Sampling & Quality Control
-
Sterile in-process sampling
-
QC / Microbiology sampling
-
Chain-of-custody closed transfer
Markets Served: Biologics ,Cell & Gene Therapy ,Vaccine MAB ,Oncology ,API ,CDMO
Cell & Gene Therapy Workflows
-
Viral vector handling (AAV, Lenti)
-
Closed small-volume transfers
-
Cryo-bag filling & aliquoting
Markets Served: Cell & Gene Therapy ,Biologics ,Oncology ,CDMO
Vaccine Manufacturing
-
Antigen preparation
-
Buffer exchange
-
Final vaccine filling lines
Markets Served: Vaccine ,Biologics ,Formulation / Fill Finish ,CDMO
Inteviz Filter Assemblies are meticulously designed in collaboration with industry experts, assembled in ISO-certified cleanrooms, and 100% integrity-tested to ensure consistent, reliable performance. Built using globally validated membranes and components, they meet the highest standards of sterility, scalability, and process compatibility. We can help accommodate your preferred filter types, membranes, and configurations—tailored precisely to your process requirements. What truly sets Inteviz apart is our end-to-end commitment—even after delivery—with comprehensive documentation, validation support, and responsive technical assistance.
|
Parameter
|
Value / Option
|
|---|---|
|
Assembly Type
|
Single-Use
|
|
Sensor Compatibility
|
Hamilton, PreSens, PendoTECH, etc.
|
|
Tubing Size Options
|
1/8” to 1” (custom on request)
|
|
Connector Types
|
Luer lock, CPC AseptiQuik, weldable
|
|
Sterilization
|
Gamma irradiated
|
|
Assembly Validation
|
100% leak tested and traceable
|
Quality Built into Every Step
From sourcing globally trusted components to final cleanroom assembly and post-production testing, every step is governed by rigorous quality protocols. Our cleanroom environment, documentation control, and traceability systems ensure our customers receive products that are safe, consistent, and compliant every single time.